Bioburden in Packaging Materials Gamma Irradiation for Sterilisation
The Risk You Can’t See in Packaging Materials
In industries such as pharmaceuticals, medical devices, and biotechnology, packaging materials are expected to protect the product from external contamination.
They are sourced from controlled environments, handled carefully, and appear clean.
But there is one factor that is often underestimated:
Bioburden in packaging materials.
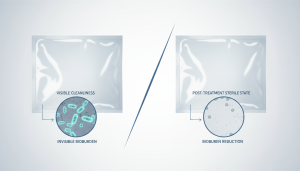
Bioburden refers to the number of microorganisms present on a material before sterilisation. And unlike visible defects, bioburden is not something you can see or assess visually.
Which means:
Packaging can look perfectly clean — and still carry microbial contamination.
A Situation Most Teams Don’t Measure Directly
In many operations, packaging materials are received, inspected, and approved based on:
- Visual checks
- Supplier documentation
- Handling practices
Everything seems in control.
But rarely is there a direct understanding of:
What is the actual microbial load on the packaging?
Because measuring bioburden is not always part of routine checks.
And that creates a silent gap.
Why Bioburden Matters in Packaging Materials
Bioburden is not just a technical parameter — it directly impacts product safety and compliance.
High or inconsistent bioburden levels can:
- Increase sterilisation requirements
- Affect sterility assurance levels (SAL)
- Impact product stability
- Lead to batch rejection
- Raise regulatory concerns
In critical applications, especially in aseptic packaging and single-use systems, this becomes even more important.
Because sterility is achieved by reducing bioburden — not by assuming it is low.
Understanding Bioburden in Real Supply Chains
Even when packaging materials are manufactured in clean environments, they go through:
- Storage
- Transportation
- Handling across multiple points
Each of these steps introduces opportunities for microbial contamination.
Environmental exposure, surface contact, and logistics conditions all contribute to the bioburden in packaging materials.
The challenge is:
This contamination remains invisible, but it is very real.
How Gamma Irradiation Reduces Bioburden
Gamma irradiation is a terminal sterilisation method that effectively reduces bioburden by inactivating microorganisms.
It uses high-energy photons (typically from Cobalt-60) that penetrate materials and disrupt microbial DNA, preventing replication.
This process:
- Reduces microbial load
- Achieves targeted sterility assurance levels
- Provides consistent and measurable outcomes
Unlike surface-level treatments, gamma irradiation penetrates through the entire packaging material, ensuring comprehensive bioburden reduction.
From Bioburden Reduction to Sterility Assurance
Sterilisation is not just about eliminating microorganisms — it is about achieving a defined level of sterility.
This is where the concept of Sterility Assurance Level (SAL) becomes important.
Bioburden plays a critical role in determining:
- Required sterilisation dose
- Process validation
- Consistency across batches
By controlling and reducing bioburden, gamma irradiation helps ensure that the desired SAL (typically 10⁻⁶) is achieved reliably.
In simple terms: lower and controlled bioburden leads to predictable sterility outcomes.
Advantages of Gamma Irradiation for Bioburden Reduction
Gamma irradiation offers several advantages when addressing bioburden in packaging materials:
- Deep Penetration
Effective for multilayer packaging and bulk materials.
- Uniform Processing
Ensures consistent reduction of microbial load across the batch.
- Terminal Sterilisation
Applied after packaging and handling, reducing post-process contamination risks.
- No Chemical Residues
A dry process with no residual concerns.
- Scalable and Reliable
Suitable for industrial-scale applications.
Validation and Control of Bioburden
In regulated industries, bioburden reduction is part of a validated sterilisation process.
This involves:
- Bioburden assessment
- Dose setting
- Dosimetry
- Process validation
- Documentation
Because sterility is not just achieved — it is demonstrated and documented.
From Assumption to Measured Control
In many cases, packaging materials are assumed to be clean and low-risk.
But without understanding or controlling bioburden, this assumption can create variability.
Gamma irradiation helps shift this approach:
From assumed cleanliness → to controlled and validated sterility.
By reducing bioburden and ensuring consistent outcomes, it provides a higher level of confidence for critical applications.
About VAPL – Gamma Irradiation Services in India
Vishvesh Agromed Private Limited (VAPL) provides gamma irradiation services in India for sterilisation of packaging materials, medical products, and industrial components.
With a focus on:
- Validation and compliance
- Quality management systems
- Process consistency
VAPL supports both domestic and global clients in achieving reliable sterilisation outcomes.
Discuss Your Sterilisation Requirements
Working with packaging materials where bioburden control is critical?
Connect with us to discuss:
- Bioburden reduction strategies
- Gamma irradiation feasibility
- Dose validation
Regulatory requirements.