Gamma Irradiation vs EO Sterilisation
Which is Better for Packaging Materials?
When it comes to sterilisation of packaging materials, two methods are most commonly considered:
- Gamma irradiation
- Ethylene oxide (EO) sterilisation
Both are widely used across industries such as pharmaceuticals, medical devices, and biotechnology. Both are validated processes. And both can achieve sterility when applied correctly.
So the natural question becomes:
Which sterilisation method is better for packaging materials?
The answer is not always black and white. It depends on the application, material compatibility, regulatory requirements, and operational priorities.
A Practical Situation Most Teams Face
Imagine you are sourcing aseptic packaging or components for a regulated application.
Everything is in place:
- Approved vendors
- Defined specifications
- Quality checks
But when it comes to sterilisation, the decision often comes down to:
Gamma irradiation vs EO sterilisation — which route should we take?
This is where understanding the differences becomes critical.
Because the choice of sterilisation method impacts not just sterility, but also material behaviour, compliance, and downstream processes.
Understanding Gamma Irradiation for Packaging Materials
Gamma irradiation for packaging materials is a terminal sterilisation method that uses high-energy photons (typically from Cobalt-60) to destroy microorganisms.
The radiation penetrates deep into the material, disrupting microbial DNA and preventing replication.
This makes gamma irradiation particularly suitable for:
- Multilayer packaging materials
- Aseptic bags and flexible packaging
- Single-use systems
- Pre-packed and sealed products
Since it is a dry process, gamma irradiation does not leave chemical residues.
Understanding EO Sterilisation for Packaging Materials
EO sterilisation (ethylene oxide sterilisation) is a chemical sterilisation method that uses gas to eliminate microorganisms.
It is typically carried out in controlled chambers, where EO gas penetrates the material and disrupts microbial activity.
EO sterilisation is often used for:
- Heat-sensitive materials
- Complex geometries
- Certain medical devices and packaging systems
However, EO sterilisation requires:
- Controlled humidity
- Aeration cycles to remove residual gas
- Strict monitoring of residual levels
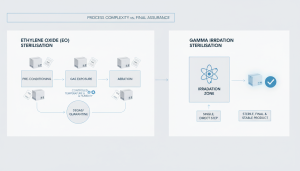
Gamma Irradiation vs EO Sterilisation: Key Differences
When comparing gamma irradiation vs EO sterilisation, several factors should be considered:
- Residues and Post-Processing
Gamma Irradiation
- No chemical residues
- No aeration required
EO Sterilisation
- Residual EO and by-products may remain
- Requires aeration time to reduce residues
👉 For applications where residual chemicals are a concern, gamma irradiation offers an advantage.
- Process Type
Gamma Irradiation
- Physical process
- Sterilisation through radiation
EO Sterilisation
- Chemical process
- Involves gas exposure and reaction
👉 This difference becomes important for material compatibility and regulatory acceptance.
- Penetration and Packaging Configuration
Gamma Irradiation
- Deep penetration
- Suitable for bulk and dense packaging
EO Sterilisation
- Gas penetration depends on packaging design
- Requires controlled permeability
👉 Gamma irradiation is often preferred for sealed and multilayer packaging materials.
- Processing Time
Gamma Irradiation
- Relatively faster processing
- No aeration phase
EO Sterilisation
- Requires multiple stages:
- Conditioning
- Sterilisation
- Aeration
👉 EO cycles can be longer due to post-sterilisation aeration requirements.
- Environmental and Safety Considerations
Gamma Irradiation
- No chemical emissions during process
- Controlled radiation environment
EO Sterilisation
- EO gas is hazardous and regulated
- Requires strict safety controls
👉 Increasing regulations around EO emissions are influencing industry choices.
Material Compatibility: A Key Decision Factor
One of the most important considerations in choosing between gamma irradiation and EO sterilisation is material compatibility.
Gamma irradiation may affect certain polymers at higher doses, while EO sterilisation may interact with materials differently due to moisture and gas exposure.
Common packaging materials such as:
- Polyethylene (PE)
- Polypropylene (PP)
are often compatible with gamma irradiation within validated dose ranges.
However, every application requires:
- Testing
- Validation
- Performance evaluation
Because ultimately:
The goal is not just sterility — it is functional performance after sterilisation.
Regulatory and Validation Considerations
Both gamma irradiation and EO sterilisation are accepted and validated methods under global standards.
However, each requires:
- Process validation
- Dosimetry or gas monitoring
- Documentation and traceability
In regulated industries, sterility must not only be achieved but also demonstrated through validated processes.
So, Which is Better for Packaging Materials?
There is no universal answer.
However, for many packaging applications — especially those involving:
- Aseptic systems
- Flexible packaging
- Single-use components
gamma irradiation for packaging materials is often preferred due to:
- No chemical residues
- Terminal sterilisation capability
- Deep penetration
- Simpler processing flow
At the same time, EO sterilisation remains relevant for:
- Heat-sensitive materials
- Specific product requirements
The choice should always be based on:
- Material compatibility
- Application requirements
- Regulatory considerations
Making the Right Choice
Choosing between gamma irradiation vs EO sterilisation is not just a technical decision — it is a risk management decision.
It involves understanding:
- Where contamination can occur
- How sterility is ensured
- What happens after sterilisation
Because in real-world operations:
Sterility is not just about the process — it is about confidence in the final product.
About VAPL – Gamma Irradiation Services in India
Vishvesh Agromed Private Limited (VAPL) provides gamma irradiation services in India for sterilisation of packaging materials, medical products, and industrial components.
With a focus on:
- Validation and documentation
- Quality management systems
- Global compliance
VAPL supports both domestic and international clients in achieving reliable sterilisation outcomes.
Discuss Your Sterilisation Requirements
Evaluating gamma irradiation vs EO sterilisation for your packaging materials?
Connect with us to discuss:
- Feasibility assessment
- Material compatibility
- Dose validation
Regulatory requirements.